|
Electricity is the movement of electrons between atomsĮlectrons usually remain a constant distance from the atom's nucleus in precise shells. For example, every atom of hydrogen (H) has one proton and every atom of carbon (C) has six protons. The Periodic Table of Elements shows elements with their atomic numbers-the number of protons they have. An element is a substance consisting of one type of atom. The number of protons in an atom determines the kind of atom, or element, it is. The neutrons carry no charge, and their number can vary. An atom is in balance when it has an equal number of protons and electrons. The positive charge of the protons is equal to the negative charge of the electrons. Protons have a positive charge (+) and electrons have a negative charge (-). The protons and electrons of an atom are attracted to each other. Electrons are held in their shells by an electrical force. The electrons would be on the surface of the bubbles, constantly spinning and moving to stay as far away from each other as possible. If the naked eye could see an atom, it would look a little like a tiny cluster of balls surrounded by giant invisible bubbles (or shells). If the nucleus was the size of a tennis ball, the atom would be the size of a sphere about 1,450 feet in diameter, or about the size of one of the largest sports stadiums in the world. 
Electrons spin around the nucleus in shells. The nucleus is made up of particles called protons and neutrons. 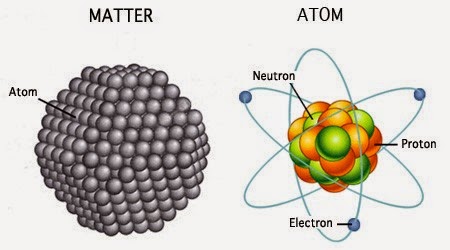 
The center of an atom is called the nucleus. Atoms are so small that millions of them would fit on the head of a pin. Everything in the universe is made of atoms-every star, every tree, and every animal. Atoms are the building blocks of the universe. To understand electricity, some basic information about atoms is helpful.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
